Raw Material Safety in the Sports Nutrition Supplements Category: What Suppliers Should Know
August 4, 2025

Safety of sourcing materials is now a critical focus in B2B supply chains. Consumers and business partners alike expect consistency, transparent sourcing, and full compliance with regulatory requirements. Any misstep can damage a company’s reputation and result in fines or product recalls.
This article outlines key steps and strategies for suppliers to meet international requirements, minimize risks, and strengthen client trust.
Key Requirements for Raw Material and Ingredient Safety
Demand for high-quality, safe products in the Food and Sports Supplements sector continues to rise. According to Grand View Research, the global dietary aids market surpassed $177 billion in 2023 and is expected to grow at an average annual rate of 9.1% through 2030.
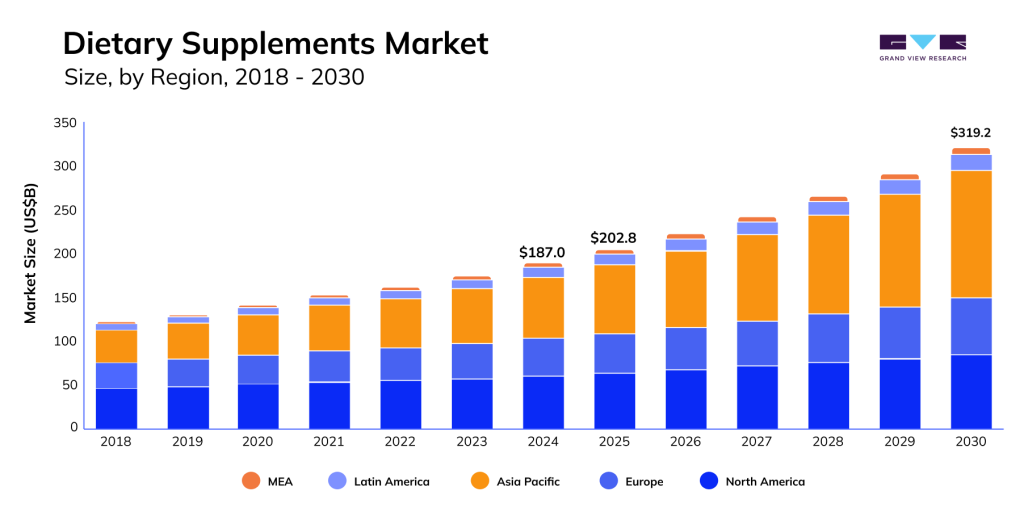
This is especially critical in sports nutrition supplements, where even minor contamination or failure to match regulatory expectations can harm a brand’s reputation and lead to fines or product recalls.
Here are the main requirements providers and manufacturers should follow to guarantee the integrity of raw inputs.
Screen for Banned Substances
In Europe, regulations under the EU Food Supplement Directive set clear rules on permitted ingredients, dosages, and processing aids in sports nutrition products. The European Food Safety Authority (EFSA) and the European Commission review and update these lists annually based on current scientific research.
This means suppliers must closely monitor not just raw materials, but also each stage of processing and storage to prevent the presence of banned or uncertified substances.
To ensure nutraceutical safety, manufacturers should:
- Implement internal quality control systems
- Stay informed on current EFSA supplement regulations
- Test every batch for compliance
- Maintain complete documentation for audits and inspections by partners or regulators
These measures help minimize risk, avoid penalties, and maintain trust across the supply chain.
Get Certified and Prove Reliability
For many importers, manufacturers, and distributors, sourcing only GMP-certified supplements is a baseline requirement. Certifications such as GMP, HACCP (HACCP supplement manufacturing), and ISO 22000 supplements are no longer viewed as formalities — they serve as essential proof that production takes place under safe, controlled conditions.
Holding these certifications gives distributors a distinct advantage and strengthens their position in the B2B market. Distributors and brands often prioritize partners with internationally recognized qualifications.
Beyond the basic standards, FSSC 22000 certification is becoming increasingly popular. It covers the entire nutritional ingredient production and supply chain. For suppliers, this certification offers an opportunity to demonstrate a comprehensive approach that includes:
- Raw material quality control
- Compliance with hygiene requirements
- Safe storage, transportation, and labeling
Regularly renewing certifications and passing audits provide a significant advantage when negotiating with new B2B partners. This fosters trust in the supplier’s brand and lowers the risk of contract refusals or terminations due to non-compliance with the EU Food Supplement Directive.
Ensure Transparency and Track the Supply Chain
Supplement traceability is a crucial factor for companies in the Food & Sports Nutrition sector. According to BCC Research, the global market for traceability systems is expected to grow by an average of 10% annually. This reflects increasing demand for transparency and control throughout every stage of the supply chain.
Assurance of components alone is not enough. Business partners expect detailed documentation that clearly shows:
- The origin of each batch
- When and by whom it was produced or processed
- Which quality certificates the batch holds
- Whether it has undergone additional lab testing
A transparent supply chain is a strong marketing advantage. For many B2B clients, it’s clear proof that the supplier meets the highest EU supplement safety standards.
What Packaging Requirements You Should Meet
Packaging is crucial for maintaining compliance and ensuring regulatory alignment. All sourcing must be food-grade, approved for direct contact with consumable products, and meet EU regulations. This applies to both primary packaging (containers, pouches, jars) and secondary packaging used for transportation.
Vendors should pay special attention to labeling. According to the European Commission, incorrect or incomplete label information is one of the most common reasons for product recalls. This can include inaccurate ingredient lists, missing language compliance, or misleading health claims that violate the EU Food Supplement Directive.
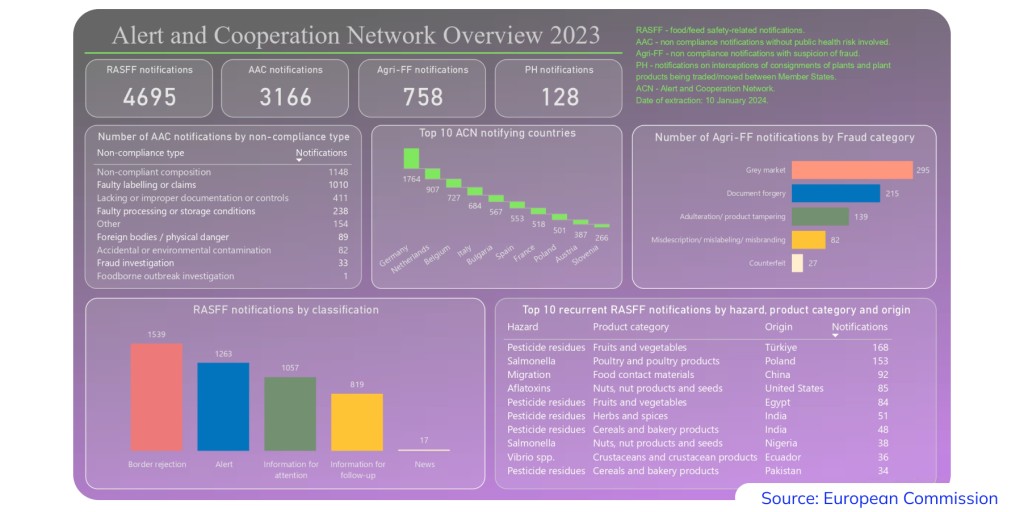
To avoid fines and potential contract losses, suppliers must ensure that all mandatory information is clearly stated on the label, including:
- Accurate list of ingredients
- Country of origin of the inputs
- Production date and expiration date
- Usage instructions and recommended daily intake
- Contact details of the manufacturer or importer
More and more B2B partners expect greater transparency, such as QR codes for certificate verification, unique batch numbers, and links to product testing results. These features help build trust and simplify supply chain audits.
Market Direction: Trends for 2025–2026
The Wellness Products sector rapidly adapts to evolving consumer demands, often introducing new market rules that raw material suppliers must soon follow.
-
-
-
-
- Clean label supplements — products free from artificial colors, preservatives, and unnecessary additives—are becoming standard and increasingly required by major brands. According to the European Clean Label Trends report by Innova Market Insights, 53% of European consumers see clear and transparent ingredient labeling as a decisive factor in their purchasing decisions. This means producers must thoroughly verify and document the composition of every batch.
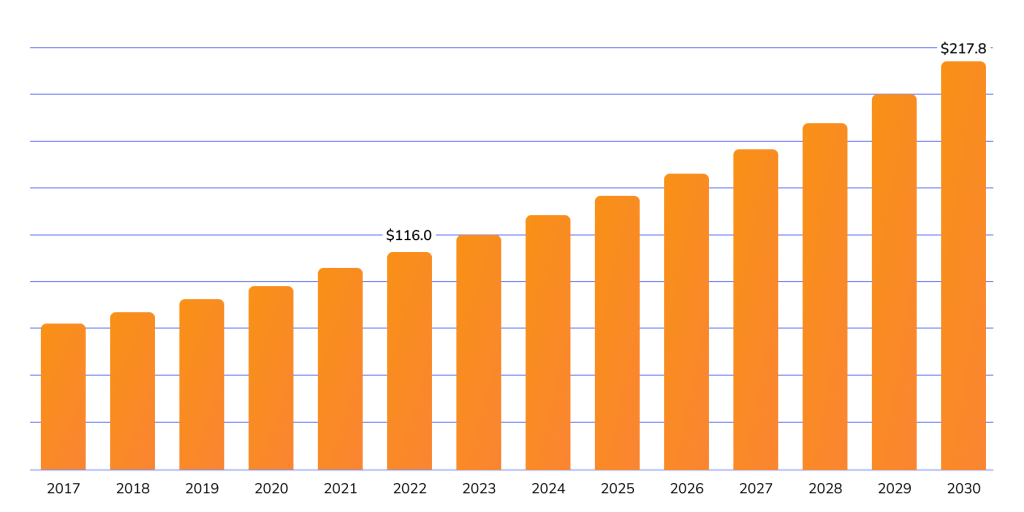
- According to Grand View Research the vegan protein powder market in Europe is projected to grow by 8.2% annually, reaching $217.8 million by 2030. Rising demand for plant-based proteins is driving higher expectations for suppliers — partners seek certifications confirming products contain no animal-derived ingredients.
- More consumers are choosing non-GMO supplements and sugar-free supplements, influenced by health-conscious lifestyles and specific dietary needs. These qualities are increasingly seen as essential in sports nutrition offerings. Distributors must be ready to provide relevant non-GMO certifications and lab reports verifying sugar content—or its absence.
Producers and traders who respond early to these trends gain a competitive edge in securing major B2B contracts by meeting the market’s increasing demand for natural products and high quality.
Building Brand Trust Through Transparency
Trust in B2B relationships goes beyond certificates and formal audits. It develops through open communication, clear collaboration guidelines, and a commitment to sharing information about the origin, reliability, and processing of ingredients.
To maintain this trust, stay connected with your partners by clearly explaining each stage of production and logistics. Provide detailed information about batches, certificates, standards, and terms of cooperation. Keep your data up to date and demonstrate openness to audits and ongoing communication.
One practical way to demonstrate openness is by using modern marketplaces that emphasize transparent and secure transactions. ServiCom is an example of such a platform. It connects verified manufacturers, distributors, and service providers, simplifies B2B contract processes, and automates documentation and delivery, making the entire supply chain more transparent and manageable. For businesses, this provides an opportunity to expand markets, attract new clients, and build lasting partnerships based on trust.
-
-
-










